This article was originally published on medium.com on June 7, 2020.
Reading time: 27 min
Disclaimer
This article is based on my research of information from publicly available sources and my personal experience. I’m not an MD or PhD in molecular biology but have a curious mind to figure things out for myself and have good analytical skills (PhD in Economics and former McKinsey consultant). The information, opinions, and references provided in this article are for informational purposes only. This article is not a medical advise and is not intended to treat, diagnose or prescribe for any illness or condition. Please consult your doctor or healthcare provider for your specific diagnosis and treatment.
Introduction
This is the 5th article in the Longevity Principle series. In this article, we deep-dive into Longevity principle #3 looking at the what, the why, and the how of mitochondrial health to improve cellular energy:
- The “What”: what we want for longevity
- The “Why”: what is often wrong and why this happens
- The “How”: what you could practically do and how it minimizes aging

This longevity principle is also described in a concise way in Google Slides below. This article offers more details and explanation.
Summary
This article is fairly scientific and may require effort to read, especially for someone new to the topic. However, in a nutshell, there are only a few things to know:
- Cellular energy is generated by mitochondria. Each cell has hundreds to thousands of mitochondria depending on cell type.
- To improve cellular energy you want to:
— Maximize the number of mitochondria. The more mitochondria each cell has, the less stress each mitochondrion will experience when satisfying cell’s energy needs. The topic of growing new mitochondria is topic is worth a separate discussion and I will talk about in detail in the Longevity Principle #4.
— Maximize energy generated by each mitochondrion. Low mitochondrial energy production creates a vicious cycle: low mitochondrial energy production -> more work for each mitochondrion to satisfy cell’s energy needs -> mitochondrial damage -> even lower mitochondrial energy production -> more damage…
— Minimize the number of damaged mitochondria. When cell reaches a certain threshold of percent of damaged mitochondria to total mitochondria, it commits a suicide called apoptosis. So fewer damaged mitochondria is better. - While all this sounds nerdy, practically, it translates into a few simple things across nutrition, supplements, accessories, physical activity:

Let’s jump into how each of these recommendations contributes to mitochondrial health.
The “What”: what we want for longevity
To understand what cellular energy is and what is needed to maximize it, we have to look into how cells produce energy, with focus on mitochondria. So, buckle up for a quick refresher on a high-school biology class. I promise it will be short.
What are mitochondria?
Unless you live under a rock, you certainly heard about mitochondria — the powerhouses of the cells. This article would not be enough to cover the topic of mitochondria in depth and I will not attempt to do it. However, it is important to spend a few minutes describing what mitochondria are and how they function.
Mitochondria (plural form of mitochondrion) represent an exciting, controversial, and complex topic. The engineering beauty of mitochondria is what sparked my initial interest in learning the science of health and longevity. Below, I want to explain why they are so exciting, controversial, and complex.
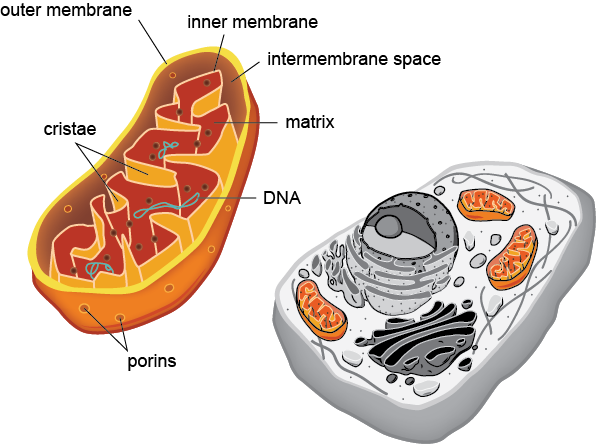
Schematic illustration of eukaryotic cell and key parts of mitochondria. Source: shmoop.com
Mitochondria are tiny organelles inside cells that use oxygen to convert food (glucose, amino acids, fatty acids) into energy (ATP or Adenosine Triphosphate). There are ten million billion mitochondria in the human body (~10% of the human body weight). There are hundreds to thousands of mitochondria in each cell depending on the type of cell:
- On average, there are 300–400 in every cell.
- Metabolically active cells (liver, heart, muscles, and brain) have thousands of mitochondria.
- Oocytes (egg cells) have ~100.000 mitochondria; infertility is related to mitochondrial dysfunction in oocytes.
Mitochondria generate ~90% of our energy and they produce energy EXTREMELY effectively.
Gram per gram, even when sitting comfortably, you are converting 10 000 times more energy than the sun every second (!!!) It’s worth rereading this sentence a few times to fully appreciate it.
Mitochondria produce energy so efficiently that they are believed to be the single reason why complex life on earth became possible. Mitochondria are what enabled organisms to grow larger while keeping energy demands low.
But why are mitochondria so powerful? To understand that we need to look deeper into how cellular energy is produced.
What is cellular energy?
Most of us think of energy in terms of calories. One calorie is defined as the amount of heat needed to raise the temperature of 1 gram of water by 1 °C. In molecular biology, energy is defined in terms of ATP consisting of nitrogenous base (adenine), a ribose sugar, and three serially bonded phosphate groups. ATP release energy when they are converted back into ADP (which has two phosphate groups) when one phosphate group is broken off. 1 ATP has ~12,000 calories (12 kcal).
How is cellular energy produced?
Normally, cellular energy can be produced aerobically (with oxygen) and anaerobically (without oxygen).
Anaerobic respiration consists of glycolysis (which can be both aerobic and anaerobic) and fermentation. We are all familiar with fermentation — the by-product of lactate fermentation is lactic acid, the one that causes the muscle burn feeling. Anaerobic respiration is a less preferred pathway because it is inefficient. It produces only 4 ATP molecules per glucose molecule (2 ATP during glycolysis and 2 ATP during lactic acid fermentation) compared to 36–38 ATP during aerobic respiration.
Aerobic respiration is more efficient as it uses oxygen. It consists of four steps: glycolysis, pyruvate oxidation, Krebs Cycle, and the Electron Transport Chain. See a graphical depiction below. Pyruvate oxidation is not always shown as a separate step as it doesn’t directly produce ATP but contributes electrons for the Electron Transport Chain.
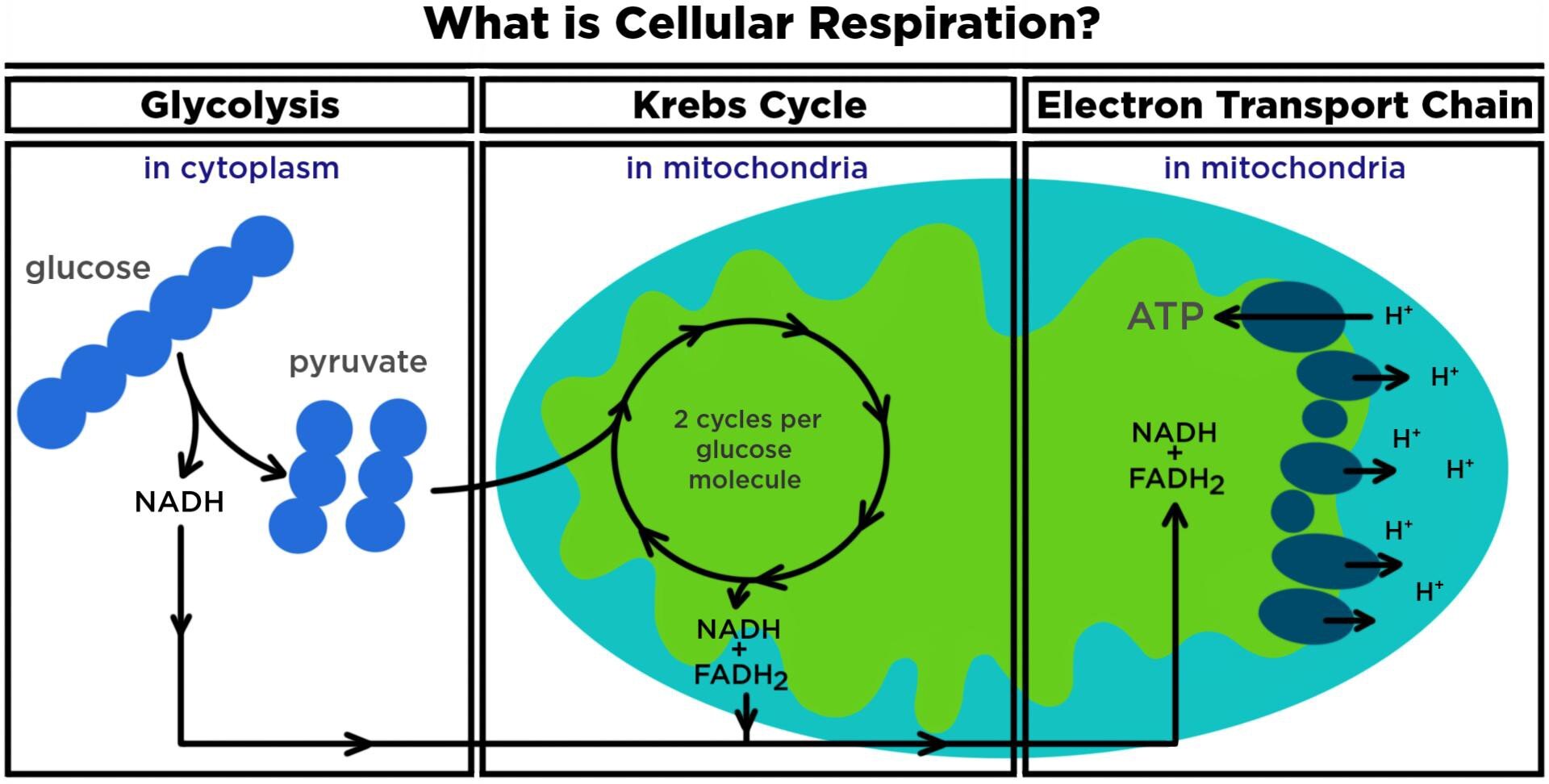
In total, aerobic cellular respiration produces approximately 36–38 ATP per glucose molecule in these 4 steps:
- Glycolysis which happens in the cytoplasm. It gives a net output of 2 ATP (produces 4 ATP and uses 2 ATP in the process) and contributes 4–6 ATP to the Electron Transport Chain (ETC) via two electron carriers called NADH.
- Pyruvate Oxidation which happens in the mitochondria. This step is not shown on the picture above as it’s a short step but an important one. In the presence of oxygen, pyruvate is converted into Acetyl CoA (an input for the Krebs Cycle) also releasing two NADH molecules contributing 6 ATP to the ETC. Pyruvate Oxidation does not directly release ATP.
- Krebs Cycle which happens in the mitochondrial matrix. It directly gives 2 ATP (produces 4 ATP and uses 2 ATP in the process) and contributes 22 ATP to the ETC via 6 NADH molecules (18 ATP) and 2 FADH₂ molecules (4 ATP).
- The Electron Transport Chain occurs in the mitochondrial membrane and it produces 32–34 ATP thanks to a contribution of 10 NADH and 2 FADH₂ molecules from the previous steps.
The Electron Transport Chain is located in mitochondria and produces 80%+ of ATP (cellular energy). Thus, focusing on mitochondrial health and efficient functioning of the Electron Transport Chain is one of the keys to improving cellular energy.
What is the Electron Transport Chain?
Zooming into the Electron Transport Chain is interesting because it helps both appreciate the evolutionary significance of mitochondria and understand the basis of health & longevity interventions to improve mitochondrial health.
The Electron Transport Chain is a series of proteins and organic molecules, most of which are organized into four large complexes labeled I to IV.
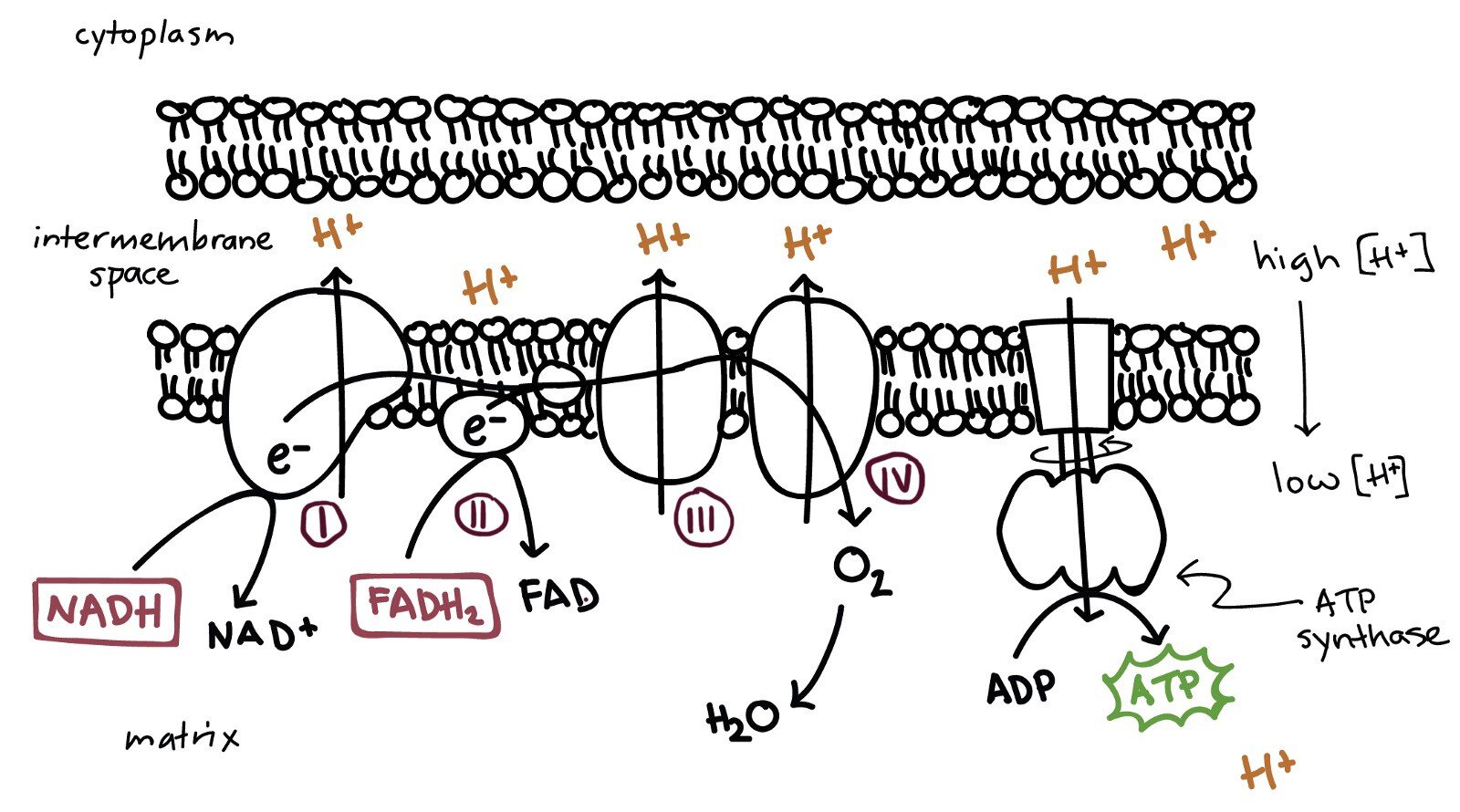
Electrons (e–) pass down the chain from complex I to complex IV. The energy released at each step moves a proton across the inner membrane to the intermembrane space. This creates a difference in proton concentration — a higher concentration of hydrogen ions (H+) in the intermembrane space and a lower concentration in the matrix. This is called a proton gradient. Hydrogen ions want to move down to a lower concentration in the matrix. They do this by going through a channel called ATP synthase, a rotor built in the inner mitochondrial membrane. Hydrogen ions moving through ATP Synthase rotate it, catalyzing the addition of a phosphate to ADP (with two phosphate molecules) to form ATP (with three phosphate molecules). ATP Synthase rotates 100+ times per second. Each mitochondrion has 300k+ ATP Synthase complexes according to some estimates.
The beauty of ATP Synthase, a rotor built in the inner mitochondrial membrane
The question remains — why are mitochondria so energy efficient? The reason is because mitochondria have an ingenious engineering solution. As we just learnt, a mitochondrial membrane is used for generating energy (i.e., pumping H+ ions through ATP Synthase) so in order to generate more energy a larger mitochondrial membrane surface area is needed. Increasing mitochondrial size wouldn’t help because that will also increase energy requirements. To solve that, mitochondria have an inner membrane that is folded many times inside the outer membrane (refer to the picture at the beginning of this article). This way, mitochondria are able to stay small but highly energy efficient — the larger the inner membrane surface area, the more ATP Synthase complexes can be placed there.
To reiterate the statement from above, mitochondria are extremely efficient at producing energy and believed to be the single reason why complex life on earth became possible. The evolutionary invention of the Electron Transport Chain is one of the reasons why that’s the case.
What are the key functions of mitochondria?
In addition to generating cellular energy, mitochondria serve other important functions. They regulate apoptosis (programmed cell death), inflammatory response, and various metabolic tasks such as calcium signalling, steroid synthesis, and hormonal signalling.
Mitochondria decide if a cell should live, grow or die.
To do that, mitochondria have their own genes called mitochondrial DNA (mDNA). mDNA is inherited from your mother because it enables more efficient nuclear-mitochondrial DNA selection in the early periods of embryo development. DNA was retained in the mitochondria, instead of being transferred to the nucleus during the evolution, because:
- it allows mitochondria to respond quickly to change in energy demands (each daily activity such as running, sleeping, reading, etc. requires- energy in different amounts).
- it gives an address tag to specific mitochondria in case repairs, chemical compounds, etc. are needed.
What are free radicals?
A free radical, aka reactive oxygen species (ROS), is a molecule with an unpaired electron on its outer shell. Mitochondria generate free radicals which attack our genes 10.000 to 100.000 times a day. While excess free radicals leakage causes DNA damage, free radicals also play various important signaling functions in mitochondria. Living organisms keep a balance between ROS and antioxidant defenses including exogenous (i.e., external) and endogenous (i.e., internal) antioxidants, such as vitamin C, vitamin E, glutathione, superoxide dismutase (SOD), catalase (CAT), and peroxiredoxins (PRXs).
Contrary to a popular belief, higher antioxidant levels do not help prevent free radical leakage and do not extend maximum lifespan. This is where one of the major controversies about mitochondria lie. However, supplemental antioxidants may correct dietary deficiencies and protect against certain diseases.
The above explanations are simplified on purpose and may miss a few interesting and important points. But going deeper would be beyond the scope of this article. If you’re fascinated by mitochondria and want to learn more, “Power, Sex, Suicide” by Nick Lane is a good place to start.
Putting it all together
As discussed in the 2nd article of this series, to promote longevity we need to:
- Maintain mitochondrial redox potential, i.e. a 50:50 ratio of oxidized and reduced electrons.
- Maintain healthy apoptosis so that only the right (old, worn-out or ill-functioning) cells are removed.
- Decrease endogenous DNA damage to avoid errors in DNA replication.
From a mitochondrial health perspective, this means:
- Preventing excessive leakage of electrons and protons (Reactive Oxygen Species or ROS) from the Electron Transport Chain. This helps prevent endogenous damage to the mitochondrial DNA, the lipid mitochondrial membrane, and enzymes and maintains a proper redox balance.
- Maintaining enough molecules to support cellular respiration and the Electron Transport Chain and its complexes I to IV (e.g., CoQ, aka Coenzyme Q10, carrying electrons from complexes I and II to complex III). This also helps endogenous damage to the mitochondrial DNA, the lipid mitochondrial membrane, and enzymes and maintains a proper redox balance.
- Maintaining a high percentage of healthy mitochondria in a cell to support high levels of ATP production. The proportion of damaged mitochondria and the ATP levels in the cell are related. When the ATP levels fall below a particular threshold (which differs for different types of cells), the cell commits cell death — either apoptosis or necrosis depending on ATP levels (necrosis may be even worse as it leads to an inflammatory response).
- Maintaining a healthy interaction among mitochondria by maintaining a flexible extracellular matrix (ECM) and dynamic cytoskeleton. The ECM is a network consisting of large molecules (mostly collagen) that provide structural and biochemical support to surrounding cells. The cytoskeleton is a structure which gives the cell its shape, helps organize the cell’s parts, and provides a basis for movement and cell division. Because mitochondria form a dynamic network inside the cell (constantly splitting (fission) and combining (fusion) among each other), they depend on a dynamic cytoskeleton and, thus, on a flexible extracellular matrix because the ECM is connected with the cytoskeleton.
Putting it simply: you need to protect mitochondria to improve cellular energy to live longer.
Here is a quick chart summarizing it:

These things do not sound particularly actionable. “How can I maintain a high percentage of healthy mitochondria” — one would think. In the “Why” chapter, we will look deeper into daily choices which cause mitochondrial inefficiency. This will make it more clear, what actions we might take to return mitochondria back to being highly efficient. We will discuss it in the “How” chapter.
The “Why”: what’s often wrong and why
After thinking about mitochondria for a while (and literally visualizing cellular energy production in my dreams), I came to think of issues with mitochondrial health in three Why’s:
- The “Why” #1. Things which directly lead to leakage of free radicals.
- The “Why” #2. Things which lead to low cellular energy.
- The “Why” #3: Things which damage mitochondrial structure and disrupt mitochondrial functions.
Let’s look at each issue separately focusing on what causes it.
The “Why” #1: Things which directly lead to leakage of free radicals
As you recall from above, reactive oxygen species, aka free radicals, are important as they serve important signaling functions but excessive leakages of free radicals is bad because this leads to DNA damage.
Excessive leakage of free radicals happens for 2 main reasons:
- Inability to discharge NADH to NAD+
- Toxic chemical agents

Inability to discharge NADH to NAD+
Definition
NAD (Nicotinamide adenine dinucleotide) is a cofactor that is central to mitochondrial metabolism as well as many other broader functions such as gene expression. NAD exists in two forms: NAD+ (oxidized form) and NADH (reduced form). A constant cycling of NAD between these two forms provides the cell with a mechanism for accepting and donating electrons. During glycolysis, pyruvate oxidation, and the Krebs Cycle, the NAD+ molecule accepts electrons (to be precise, two electrons and one hydrogen ion) and becomes reduced to the NADH form. The NADH molecule then donates (i.e., loses) a pair of electrons to Complex I of the ETC. As a result, (1) the hydrogen ion (H+) is moved into the intermembrane space to create a proton gradient to rotate ATP Synthase and (2) the NAD+ molecule is formed again which goes back to be recycled to NADH again during glycolysis, pyruvate oxidation, and the Krebs Cycle.
Longevity objective
In general, a higher NAD+/NADH ratio is better for higher cellular energy. Intuitively, it makes sense — the NAD+/NADH ratio is determined by the rate at which NADH is generated (think of it as food consumed) and the rate at which NADH is reconverted back into NAD+ at Complex I of the ETC (think of it as releasing energy from the food consumed). So a higher NAD+/NADH ratio, would mean releasing more energy from less food.
Problem
The problems come from:
— consuming too much food generating elevated NADH levels. This puts increased pressure on the Electron Transport Chain to recycle larger amounts of NADH into NAD+ and produce more energy (ATP). More electron pressure is induced on Complex I.
— having a sedentary lifestyle or little physical activity not spending the energy our mitochondria produce. This reduces the rate of electron flow making Complex I more reactive and leading to increased formation of ROS.
Even under normal conditions (i.e., not overeating and exercising), Complex I of the ETC is a major source of reactive oxygen species in our cells. Combine that with overeating and lack of physical activity and you get constant excessive free radical leakage.
Down the line, as a ratio of NAD+/NADH declines further, glucose metabolism by the glycolysis pathway will be disrupted, leading to high blood sugar levels and eventually diabetes. Glucose will be diverted to the polyol pathway (which converts glucose into fructose) further increasing NADH levels and decreasing NAD+/NADH ratio. This would further increase free radical (ROS) leakage leading to chronic inflammation.
Toxic chemical agents
Definition
Exposure to different chemical agents may disrupt normal cellular respiration and cause mitochondrial damage. It makes sense given that mitochondria function via electrochemical reactions. So if you add wrong chemical agents or even the right chemical agents but in wrong amounts, they may disrupt normal electrochemical reactions.
Problem
— Consuming foods high in amino acid Tyramine (soy products, aged cheeses, smoked fish, cured meats, some types of beer) leading to oxidative damage to mitochondrial DNA.
The “Why” #2: Things which lead to low cellular energy
Cellular respiration and the Electron Transport Chain require various molecules to function properly to support passing of electrons between Complexes I and IV and continuous formation of ATP via ATP Synthase. Just to name a few: oxygen, ubiquinol/ubiquinone, glutathione, nucleotide pools. Let’s look deeper at:
- Oxygen deficit
- Reduced synthesis of Ubiquinol/ubiquinone (aka CoQ10)
- Glutathione deficiency
- Depletion of nucleotide pools

Oxygen deficit
Definition
Oxygen acts as a final electron acceptor that helps move electrons down a chain that results in ATP. Oxygen deficit means that there would be not enough oxygen to interact with ATP Synthase and ATP production would halt.
Problem
— Consuming too much iron may lead to iron overload which has been associated with oxygen deficit in mitochondria and Huntington’s disease and other issues such as tissue damage after ischemia. Animal proteins top the list so consuming a lot of red meat may cause issues with mitochondria.
Reduced synthesis of Ubiquinol/ubiquinone (aka CoQ10)
Definition
Ubiquinol helps carry electrons from the ETC complexes I and II to complex III. In the process, it gets oxidized. The oxidation of ubiquinol converts it to ubiquinone (better known as Coenzyme Q10) and results in the movement of protons H+ into the intermembrane space. This process is cyclical — it’s called the Q cycle.
Problem
— Taking medications such as statin-medications, beta-blockers, hypoglycemic (diabetic) medications, and tricyclic antidepressants leading to impaired synthesis of CoQ10.
— Being 30+ years old leading to reduced synthesis of CoQ10.
Glutathione deficiency
Definition
Mitochondria are the primary intracellular site of oxygen consumption and the major source of free radicals, most of them originating from the ETC. Mitochondria rely on antioxidants for protection against free radicals with glutathione being the most abundant and effective antioxidant. Glutathione deficiency has been associated with increased susceptibility to oxidative and nitrosative stress, decreased mitochondrial fat oxidation, and older age(perhaps not surprisingly).
Problem
— Consuming too little sulfur rich foods such as cruciferous vegetables, eggs, etc. Glutathione is synthesized using sulfur atoms, found in two essential (i.e., not synthesized in the human body) amino acids — methionine and cysteine — as well as a chemical compound called sulforaphane. While methionine and cysteine (mostly found in animal proteins) should be consumed in moderation to avoid upregulation of the mTOR pathway, sulforaphane (mostly found in vegetables) can and should be consumed in high amounts.
— Working out too little and having sedentary lifestyle. Exercise creates oxidative stress which stimulates glutathione synthesis. For example, this study has shown lower levels of glutathione in a no exercise group of people compared to exercise groups.
Depletion of nucleotide pools
Definition
Nucleotides are the basic building blocks of DNA and RNA. There are 2 groups of nucleotides: purines are the two-carbon nitrogen ring bases (adenine and guanine) and pyrimidines are the one-carbon nitrogen ring bases (thymine and cytosine). You guessed it right — nucleotides also play a crucial role in ATP (energy) synthesis — ATP consists of adenine base attached to ribose, and a chain of three phosphate groups bound to ribose.
Problem
— Not recovering from strenuous aerobic exercise (defined as over 1 hour a day for extended periods of time) which may lead to apoptosis (cell death) and an athletic heart syndrome (which in some cases leads to cardiac arrest and death). While aging due to sedentary lifestyle is probably a bigger issue than due to overtraining, it’s still important to recover. During strenuous aerobic exercise, once ATP is used, your body will combine two ADP molecules to produce ATP and AMP (one phosphate group). This AMP will be further broken down and eliminated from the cell reducing the purine pool. Overtraining also leads to reduced glutamine availability and glutamine is essential for synthesis of both purine and pyrimidines nucleotides.
— Consuming (too much) alcohol leading to NAD+ pool depletion.NAD+ is used to metabolize alcohol into acetaldehyde and further to acetate. Alcohol consumption decreases NAD+ pools. This leads to decreased respiratory rates and ATP levels, and increased production of ROS.
The ‘Why’ #3: Things which damage mitochondrial structure and disrupt mitochondrial functions
The third way we can lose mitochondrial energy and age is by inducing damage to mitochondrial structure and dynamic network and various functions such as apoptosis. While both The Why’s #1 and #2 will contribute to this so it’s hard to be perfectly MECE (mutually exclusive and collectively exhaustive), there are a few things that contribute to this more directly which I want to call out separately, specifically:
- Formation of Advanced Glycation End Products (AGEs)
- Heavy metals
- Carcinogens from incorrectly grilled meats
- Toxic molds and mycotoxins

Formation of Advanced Glycation End Products (AGEs)
Definition
Advanced glycation end products (AGEs) are modifications of proteins and fats which happen after contact with sugar. This process is called glycation. AGEs comprise 20+ types of compounds which potentially mediate a wide variety of pathological effects. AGEs are a bio-marker implicated in aging and development of chronic disease such as diabetes, atherosclerosis, and Alzheimer’s disease.
Most research on AGEs is either done in vitro (i.e., in glass) or uses worms, rats, and mice. While researching AGEs in humans is challenging because the levels of AGEs are hard measure and AGEs build up gradually, the effects of AGEs can be easily seen in most of us — in skin wrinkles and brown spots.
Less visible to an eye but visible to a blood pressure monitor is an increase in systolic blood pressure (the first number in blood pressure measurement). This happens because AGE cross-links impair the normal elasticity of heart and blood vessels leading to hardening of the arteries (blood vessels carrying oxygen-rich blood away from the heart). This can also lead to things such as bleeding stroke.
How do AGEs impact mitochondria: extracellular matrix (ECM) stiffness and cytoskeletal tension
Impact of AGEs goes far beyond mitochondria. But let’s focus on how do AGEs disrupt mitochondrial functions?
Mitochondria do not exist in isolation and are not static. They are highly dynamic organelles constantly interacting with other mitochondria going through fission (splitting) and fusion (combination). In this process, mitochondria interact with cytoskeleton of the cell.
As mentioned above, cytoskeleton is a cell structure. Generally, we want cytoskeleton to be flexible and dynamic. However, over time it becomes rigid and tense. This happens because of glycation which leads to cross-linking of collagen fibers and the extracellular matrix becoming stiff. ECM stiffness creates cytoskeleton tension which limits mitochondrial fission and fusion.
Longevity objective
What can we do about AGEs?
The first option is to break down AGE cross-links after they have been formed. No drug has been found to consistently do that in healthy (non-diabetic) humans but there has been great success in animals with Alagebrium which, unfortunately, haven’t been effective for humans. That is because Alagebrium was effective for alpha-diketones cross-links which are less common in humans than in animals (the most common AGE in humans is glucosepane). So part of the issue with this approach to AGEs is that there are many different types of AGEs. Breaking down each different type of AGE requires a specific drug. While this is challenging, drug-induced break down of AGE cross-links is a very promising area of research with many longevity scientists being excited about it.
The second option is to minimize or prevent formation of AGEs to slow down the rate of collagen cross-linking. There is a great deal of debate about the degree by which AGE formation can be slowed down. For example, Aubrey de Grey in his famous book “Ending Aging” argues that it only makes sense to focus on breaking down AGEs since the AGE formation process is believed to be somewhat random. While that is true, it’s also clear that higher blood sugar levels lead to more AGEs.
The degree by which AGE formation is reduced by decreasing blood sugar levels in humans with healthy blood sugar levels (below 90 mg/dL fasting glucose) is unclear and likely relative small. But for folks interested in maximizing chances of being healthier and living longer, it doesn’t matter that much — the best course of action is still to do what should minimize formation of AGEs. For example, it might be possible to contain glycation through a process called transglycation.
One of the good relative indicators of AGE formation is a well-known lab test called glycated hemoglobin or HbA1c. This test measures the average amount of sugar that has been present in the blood over a period of three months. HbA1c is what’s called an Amadori rearrangement product. These are not AGEs yet but may become AGEs — that’s why HbA1c is a not a fully accurate indicator of AGE formation. Still, HbA1c is a good lab test to do regularly.
Problem
AGEs can form within the body and also come from certain foods contributing to body’s AGE pool. Both problems arise from not eating right:
— Consuming too much sugars, especially fructose, leading to formation of AGEs in the bloodstream— Consuming foods high in AGEs due to cooking at high temperatures(barbecuing, grilling, roasting, baking, frying, sautéing, broiling, searing, and toasting) and long pasteurizing (aged cheeses)
— Consuming foods cooked with heat-oxidized oils (olive, sunflower, etc.) leading to formation of dicarbonyls and AGEs
Heavy metals
Definition
While it is well known that mitochondria are important targets for heavy metals, the mechanisms by which they disturb mitochondrial function are not well understood. The impact of heavy metals on mitochondria is metal-, time-, and dose-dependent — different metals produce different toxic effects at different doses and time of exposure. Some types of heavy metal exposure may actually be beneficial but it’s best not to experiment with that to be on the safe side. Overall, heavy metals exposure leads to increased reactive oxygen species formation, inhibition of the respiratory chain, rapid dissipation of mitochondrial membrane potential, DNA damage, and cell death (via both apoptosis and necrosis).
Longevity objective
We want to minimize heavy metals exposure which means minimizing consumption of heavy metals and/or clearing them from our bodies. Clearance is a separate topic which will be covered in Longevity principle 7 “Detoxify”. For now, let’s talk about consumption, specifically, sources of heavy metals.
Problem
Many of the foods and drinks we consume daily contain heavy metals:— lead: water pipes
— mercury: larger and longer lived fish
— uranium: root crops
— arsenic: brown rice
— thallium: kale
— and the list goes on
We can’t avoid heavy metals altogether because they are present in so many things. But we can be more selective when picking things — like eating salmon instead of tuna. More on it later.
Carcinogens from incorrectly grilled meats
Definition
A carcinogen is anything that promotes the formation of cancer. There are two types of carcinogens produced by grilling meat:
— HCAs (heterocyclic amines) associated with mitochondrial degeneration. HCA exposure has been associated with significantly enlarged mitochondria in rates.
— PAHs (polycyclic aromatic hydrocarbons) associated with disruption of apoptosis. Apoptosis is a controlled type of cell death (vs. uncontrolled necrosis). While apoptosis in healthy cells is bad (for example, recall from above: the induction of apoptosis by NAD+ pool depletion due to alcohol consumption), apoptosis in unhealthy cells is good. But PAHs disrupt apoptosis so that even unhealthy cells don’t commit suicide and may further replicate leading to cancer.
Problem
So how do HCAs and PAHs get formed?
— HCAs are formed when you cook muscle containing meat at high temperatures — amino acids, sugar, and creatine (found in muscle meat but not in organ meat) react and form HCA.
— PAHs are formed when fat from meat reacts directly with a heated surface or open fire causing flames and smoke. PAHs contained in this smoke then adhere to the surface of the meat.
Toxic molds and mycotoxins
Definition
Last but not least are the molds. When talking about mold toxicity, we refer to mycotoxins — toxic secondary metabolites produced by certain types of molds. There is a significant body of research on health risks associated with mycotoxins exposure. Different mycotoxins have different toxic effects ranging from DNA damage and mitochondrial membrane damage to induction of apoptosis (cell death).
Problem
— Consuming mold contaminated foods; the key types of mycotoxins are aflatoxin, ergot alkaloids, fumonisin, trichothecenes, ochratoxin, and zearalenone and the moldiest foods to avoid are corn, wheat, soy, cereal grains, peanuts.
— Exposure to environmental molds — in homes, offices, and other buildings. The Moldy is a fascinating movie on this topic.
The “How”: what you can practically do and how it minimizes aging
Based on the above, there are several key objectives that each intervention is aimed at. As always in health & longevity, different interventions will influence multiple different pathways.
Objective 1. Increase the NAD+/NADH ratio
Nutrition:
— Practice short-term and/or intermittent fasting and/or caloric restriction
— Follow keto diet but add carbs once every few days to avoid insulin resistance
Exercise:
— Weightlifting, HIIT, and/or endurance training
Supplements:
— NAD+ boosters (act as a gas pedal for SIRT genes): Thermally stabilized Oxaloacetate (benaGene or KetoPrime), Vitamin B3 (nicotinamide/niacid) or more biologically efficient Nicotinamide riboside (NR) (TruNiagen is FDA-approved) or nicotinamide mononucleotide (NMN) (no FDA-approved brands yet but powders can be found online; make sure they are tested for heavy metals)
— Consider adding “fuel for SIRT genes”: Resveratrol, Pterostilbene and/or Fisetin
Objective 2. Minimize leakage of reactive oxygen species
Nutrition:
— Avoid toxic chemical agents: foods with high amount of Tyramine (soy products, aged cheeses, smoked fish, cured meats, some types of beer)
— Consume resistant starch rich foods feeding gut bacteria producing a short-chain fatty acid, butyrate (more on this in Longevity Principle 6), improving mitochondrial respiratory capacity
Supplements:
— Carbon 60, a highly potent antioxidant; it has a range of medical applications also be used topically to reduce wrinkles; this brand is endorsed by Dave Asprey. I do not use Carbon 60 and can’t recommend any brands. Carbon 60 mimics Superoxide Dismutase (SOD), an enzyme playing a key role in protection from superoxides (one of the main types of ROS in the cell) as well as Nuclease, a key enzyme involved in DNA repair. In a 2012 study, c60 treated mice lived up to 90% longer compared to controls. However, a 2020 study failed to reproduce life extension benefits and showed potential toxicity from taking oxidized olive oil. Unless you have excessive leakage of free radicals due to a genetic mutation or radiation (e.g., from flying), c60 is unlikely to have life extension benefits (or worse, it can be toxic).
Objective 3. Enable consistent respiration and oxygen uptake
Nutrition:
— Minimize animal proteins (beef, etc.) and in “clean” form (grass-fed, etc.)
Supplements:
— Avoid iron supplements
— Add Vitamin B6 (<100 mg per day) to improve delivery of oxygen to the mitochondria
— Add L-Arginine to restore nitrous oxide levels and improve mitochondrial respiration
— Add Biotin to restore mitochondrial function
Environment:
— Consider doing blood donations
Accessories:
— Use red and near-infrared light therapy to improve oxygen uptake by enabling better pairing with Cytochrome C Oxidase. Cytochrome c oxidase is is a photoreceptor (i.e., it absorbs light protons) in our mitochondria which helps oxygen to be used efficiently by the mitochondria to power the ATP synthase pump. How does it do that? In mitochondria, there is a direct competition between Nitric Oxide (NO) and O2 for binding with Cytochrome C Oxidase. When NO binds with Cytochrome C Oxidase, it prevents O2 from binding with it which inhibits efficient ATP production. Red and near-infrared light therapy helps prevent NO from binding with Cytochrome C Oxidase, let’s oxygen in, and increases the respiration rate and ATP production. What’s great that Red/NIR light is effective for different types of cells in our body. For example, in this study, exposure to a 670 nm near-infrared wavelength led to a 20% increase in ATP levels in retina.
Objective 4. Increase glutathione levels
Nutrition:
— Eat sulfur rich foods (cruciferous vegetables, eggs, etc.)
Supplements:
— Consume undenatured grass-fed whey protein; here are my favorite cow (from Jersey cows with A2 casein) and goat whey
— Add N-Acetyl-Cysteine
— Add time-released Vitamin C
— Add R Alpha-Lipoic Acid
— Add Selenium
— Add L-Glutamine
— Add Milk Thistle
— Practice IV injections of Glutathione/Vitamin C at times of high stress (e.g., jet lag)
— Use liposomal glutathione for extra boost (but it’s expensive)
Physical Activity:
— Exercise (weightlifting, HIIT, and/or endurance training) to stimulate glutathione production but remember to recover well
Objective 5. Compensate for purine pool depletion
Supplements:
—Add D-Ribose to replenish purine pools quickly (I take 2–4 grams daily in powder form)
Objective 6. Compensate for CoQ10 deficiency
Supplements:
— Add CoQ10 (Ubiquinone, the reduced form)
— Combine with Shilajit for improved efficiency
Objective 7. Compensate or avoid possible deficiencies of other compounds supporting the ETC
Nutrition:
— Consume alcohol in small amounts only (e.g., a glass of wine) or not at all
Supplements:
— Use Creatine Monohydrate to have enough creatine phosphate to convert ADP to ATP (i.e., calories) which happens by adding a phosphate molecule and protect from oxidative stress
— Consider Vitamin B2 (Riboflavin) to improve flow of electrons in ETC
— Use Magnesium which is one of key cofactors in the production of ATP
— Add Acetyl L-Carnitine to improve transport of fatty acids across the inner mitochondrial membrane
Objective 8. Minimize the amount of AGEs
Nutrition:
— Minimize consumption of sugar, especially fructose
— Avoid foods cooked at high temperatures (barbecuing, grilling, roasting, baking, frying, sautéing, broiling, searing, and toasting) and long pasteurized (aged cheeses)
— When cooking, use the right methods: cook with moist heat, use shorter cooking times, cook at lower temperatures, use acidic ingredients such as lemon juice or vinegar, and use oils/fats with 200°C+ smoke point
— Consume free amino acids (e.g., carnosine and ansurine), polyamines (e.g., spermidine), thiols (e.g., NAC and α-lipoic acid), and thiolamines to enable transglycation. As you see, these recommendations are similar to the objective #4 but you may also add L-Carnosine.
Objective 9. Minimize intake and impact of heavy metals
Nutrition:
— Minimize intake of heavy metals (tap-water, tuna, shark, and brown rice are easy to avoid)
Supplements:
— Add heavy metal binders (chlorella, activated charcoal, and bentonite clay) to capture heavy metals — more on this in Longevity Principle 7 “Detoxify”
Objective 10. Minimize carcinogens from grilled meats
Nutrition:
— Use foil when grilling meat, marinate or pre-cook meats or grill more vegetables instead of meat to avoid carcinogens
— Do NOT grill meat on an open fire
Objective 11. Minimize exposure to mycotoxins
Nutrition:
— Avoid corn-fed animal products. Unfortunately, restaurants and offices don’t have good meat so I stick with vegetables there
— Avoid corn, peanuts, and wheat grains
— Cleaner coffee: stick with South American grades of coffee which have lowest levels of Ochratoxins, higher elevation beans, more expensive specialty grades which are less likely to have molds and/or espresso
— Cleaner nuts: soak nuts before consuming
— Cleaner chocolate: stick with European chocolate which has stricter quality standards: Vivani (find on eBay), Blanxart (find on eBay) and Montezuma (Trader Joe’s) are my favorite
Environment:
— Avoid mold in offices, homes, and your surroundings
While that sounds like a lot of complex stuff to do, most of it can be summarized in a single page:

If you want to go deeper into different nutritional components involved in mitochondrial processes, here is an excellent article and an overview picture below.
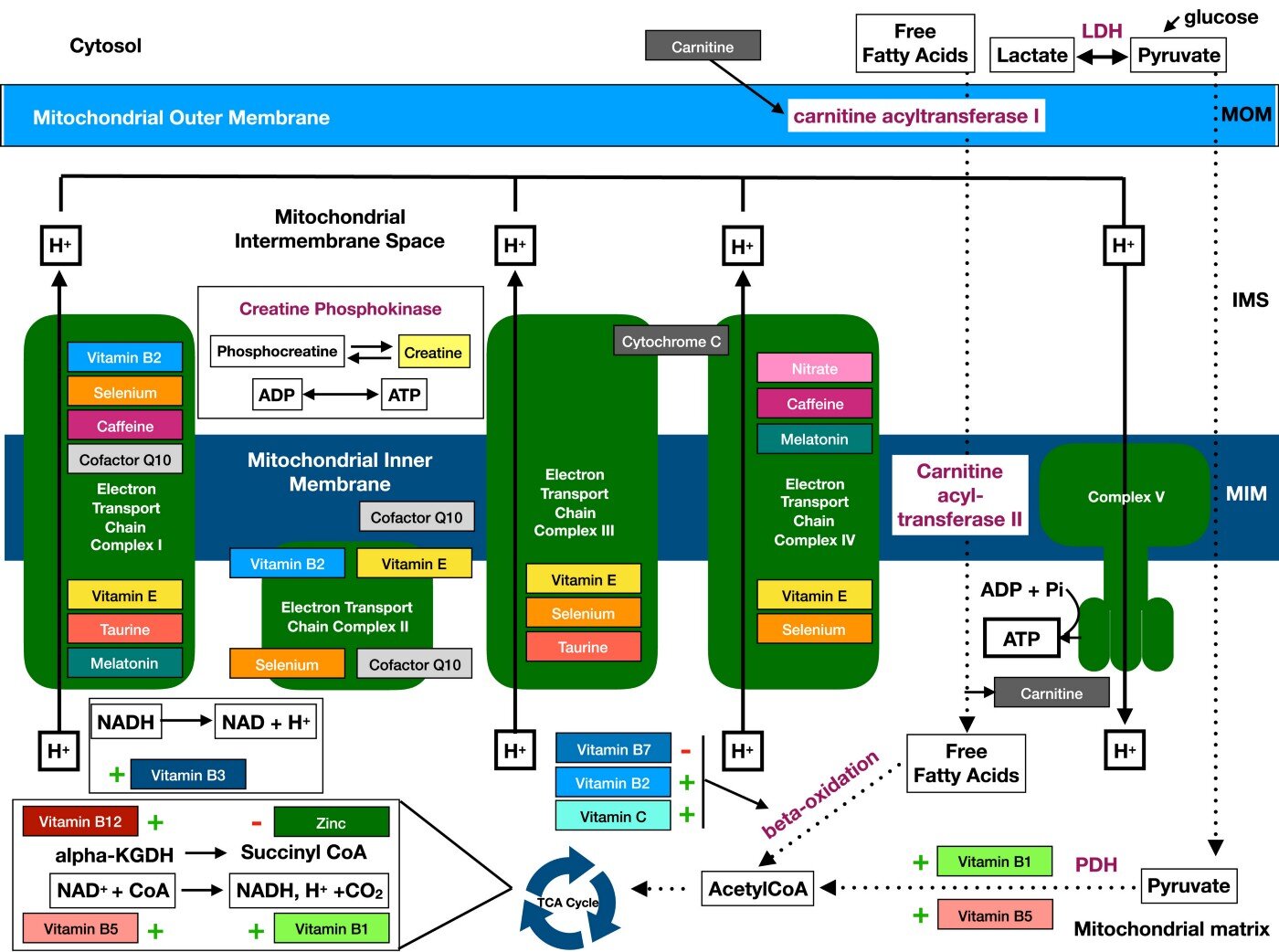
In practice
I practice red/near-infrared light therapy every other day for 30 minutes either in the morning or before bed.
There are many brands on the market. Joovv is probably the most well known endorsed by many well known people. The downside is that it gets quite expensive if you want a device powerful enough to cover a large treatment area.
I use a mat by TrueLight combined with an energy square which use 4 different wavelengths (Near-Infrared (NIR), 2 types of Red, and Yellow) penetrating tissues and boosting mitochondria on different levels. I also use this time to read which gives an additional boost to my brain cells.
Red and near-infrared light therapy has a wide number of benefits depending on length of the light waves. This book is a good place to start if you want to learn more.

