This article was originally published on medium.com on February 23, 2020.
Reading time: 16 min
Disclaimer
This article is based on my research of information from publicly available sources and my personal experience. The information, opinions, and references provided in this article are for informational purposes only. This article is not a medical advise and is not intended to treat, diagnose or prescribe for any illness or condition. Please consult your doctor or healthcare provider for your specific diagnosis and treatment.
Longevity principles series
This is the 4th article in the Longevity Principle series. In this article, we deep-dive into Longevity principle #2 looking at the what, the why, and the how of how to pause “growth mode” to live longer:
- The “What”: what we want for longevity
- The “Why”: what is often wrong and why this happens
- The “How”: what you could practically do and how it minimizes aging
TL;DR: summary recommendations
If you are only interested in practical recommendations, here they are below. Read on to learn about the recommendations in more detail and the science behind them.

The “What”: what we want for longevity
In overview of the longevity principle #1, we talked about the circadian cycle — a natural cycle repeating approximately every 24 hours which controls gene expression, molecular pathways, cells, and tissues. Humans (and other living beings) have another bigger cycle called Circannual Cycle— a self-sustained rhythm of approximately 1 year which controls major biological processes and gene expression.
The Circannual Clock is not fully deciphered yet, especially in humans, as it is difficult to study, for 2 main reasons:
- It’s beyond the scope of most PhD projects or tenure grant of funding due to a longer time-scale of the phenomenon.
- It’s difficult to dissociate the circannual clock from other developmental processes.
It’s clear, however, that the circannual cycle exists so that organisms can adapt to the Earth’s annual periodicity and respective periods of low and high energy availability.
The circannual cycle is regulated by the cyclical epigenetic regulation of chromatin structure (DNA and histone proteins). In other words, the circannual cycle is determined by gene expression and has two distinct states:
- ‘Summer’ — genes are active, i.e., growth.
- ‘Winter’ — genes are silenced, i.e., regression.
Unfortunately, for our health and lifespan, food has become widely available 365 days a year recently. So the growth genes are always active — we stay in the growth mode — which accelerates aging and increases risks of diseases such as cancer, diabetes, heart disease, and Alzheimer’s.
One of the keys to longevity and health is to pause the growth mode—or live in accordance the Circannual Cycle.
Historically, humans have been in prolonged states of seasonal energy restriction and eaten seasonal foods. That’s challenging today because:
- You need to have a truly strong will-power to put yourself in a prolonged state of “seasonal energy restriction”, for example, by restricting your caloric input to 70% of your daily needs for 6 months or more.
- Most foods are available 365 days a year so it’s hard to know which foods are truly seasonal (it may be imported from a different place where it’s seasonal).
- Most of us have a combination of genes from different geographical areas so it’s hard to know which foods are seasonal for us depending on our genetic ancestry.
However, there are 2 simple principles that can make your circannual clock function better and pause the growth mode:
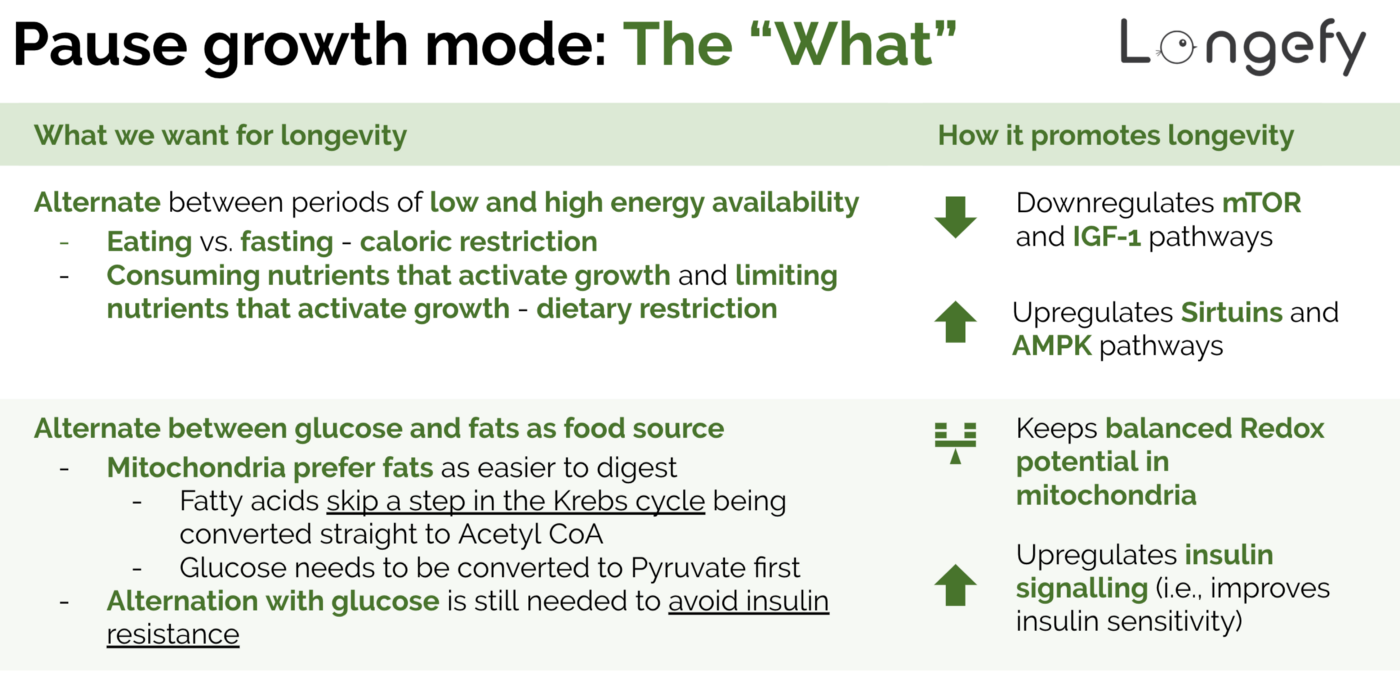
- Alternate between periods of low and high energy availability.
- Alternate between glucose and fats as food source.
Alternate between periods of low and high energy availability
First of all, let’s look at why having periods of low and high energy is important — how it promotes longevity. The state of energy availability influences several key biological pathways, most importantly mTOR, IGF-1, AMPK, and SIRT1.
mTOR or the mammalian target of rapamycin
- Definition: mTOR consists of 2 complexes — mTORC1 and mTORC2. Most of the research has been done on mTORC1 — a nutrient sensing (i.e., it is inhibited or expressed based on nutrient availability) kinase (an enzyme that adds phosphate groups to other molecules) regulating cell growth and protein synthesis. mTORC2 is regulated by growth factors though this complex has been less studied.
- Relationship to longevity: mTOR inhibition has been shown to extend lifespan in yeast and animals. While it’s much harder to show if mTOR inhibition leads to increased lifespan in humans because it’s hard to run an experiment, mTOR activation has been shown to be associated with advancing age in humans.
- Best ways to inhibit/downregulate: dietary restriction (consuming less growth amino acids — methionine, cysteine, and isoleucine — which activate mTOR and are prevalent in red meats) and caloric restriction (consuming fewer calories, short-term fasting, intermittent fasting/time-restricted eating).
IGF-1 or insulin like growth factor
- Definition: IGF-1 is a hormone similar to insulin (that’s why it’s called insulin like) which sends growth signals to your cells. IGF-1 is closely related to a more commonly known HGH (human growth hormone) with most of the HGH effects being mediated through IGF-1.
- Relationship to longevity: it has been shown in both animal and human studies that reduced IGF-1 levels/signaling lead to improved longevity and lower risk of many cancers. In humans, it has been shown that centenarians (people older than 100 years old) have low circulating IGF-1 bioactivity and that mutations in IGF-1R gene leading to reduced IGF-1 signaling are overrepresented in centenarians compared to the younger control subjects. However, relationship between IGF-1 and longevity is not as clear as black and white. First, IGF-1 levels naturally decline with age so lower levels of IGF-1 don’t necessarily stop aging but often indicate it. At the same time, it’s been shown that lower IGF-1 levels in mice are associated with longevity. Second, IGF-1 production is stimulated by exercise via growth hormone (GH) secretion. Given that regular exercise is associated with longevity, we shouldn’t stop exercising. Though it’s a good idea to avoid IGF-1 activation by exogenous sources.
- Best ways to inhibit/downregulate: dietary restriction (consuming less exogenous IGF-1 which are prevalent in cow’s milk and milk products) and caloric restriction.
AMPK or AMP-activated protein kinase
- Definition: AMPK is an enzyme that acts as a sensor of the cellular energy state, at both the cellular and whole-body state, based on the changes in intracellular AMP levels or external cues such as nutrients and hormonal signals.
- Relationship to longevity: AMPK activation has been linked to longevity in multiple studies. AMPK decline with aging leads todecreased autophagy, increased oxidative stress, increased inflammation, increased fat deposition, and other issues. AMPK is closely linked with the broader anti-aging signaling network including mTOR and IGF-1. For example, AMPK can inhibit the activity of mTORcomplex promoting longevity. AMPK and IGF-1 signaling pathways are mutually inhibitory: the IGF-1 pathway can repress many targets of AMPK signaling and AMPK can inhibit IGF-1 signaling.
- Best ways to activate/upregulate: modulate a state of energy deficit through caloric restriction and/or use of drugs/supplements mimicking caloric restriction (metformin, berberine, oxaloacetate, etc.).
Sirtuin 1 or Silent information regulator 1
- Definition: SIRT1 is a protein that acts as a sensor of the cellular energy state based on the levels of NAD+, a co-factor that is central to metabolism being involved in redox operations (carrying electrons from one operation to another). SIRT1 is the most studied enzyme from a broader family of Sirtuins (SIRT1–7). As David Sinclair describes in his excellent book “Lifespan: Why We Age and Why We Don’t Have To”, the sirtuins in mammals have evolved to carry many functions such as DNA stability, DNA repair, cell survivability, metabolism, and cell-to-cell communication.
- Relationship to longevity: Sirtuin 1 is best known for its role in mediating the increase in longevity in various species, including yeast, worms, and possibly mammals. As we age, NAD+ availability declines as shown in many species including mice and humans. This reduces sirtuin activity and affects the communication between the nucleus and mitochondria at a cellular level and also between the hypothalamus and adipose tissue at a systemic level. It’s worth noting that SIRT1 hasn’t been well studied in humans yet (only on human cells).
- Best ways to activate/upregulate: indirectly by increasing NAD+ levels which act as the fuel for SIRT1 and directly by activating the SIRT1 gene which act the gas pedal. This means exercise, caloric restriction, dietary restriction, hormesis (beneficial stress), and sleep for both NAD+ and SIRT1 and specific drugs/supplements for NAD+ and SIRT1. Interestingly, SIRT1 and AMPK has been shown to upregulate each other.
The above mentioned longevity pathways are all nutrient sensing pathways which are downregulated or upregulated by the presence of nutrients. The secret to improving health and lifespan is to cycle periods of growth and regression by controlling the nutrient intake — looking at what and how much you eat — and the energy spend — how much energy you use.
Once again, cycling between growth and regression is key. For example, you don’t want to completely suppress mTOR because it regulates muscle growth and moderate muscle mass is associated with longevity. At the same time, you don’t want to overexpress mTOR by eating too much animal foodsbecause it leads to aging, oxidative stress, and inflammation. So you need to alternate between:
- Eating and not eating (fasting). This is called caloric restriction — how much you eat.
- Consuming nutrients that activate growth and limiting nutrients that activate growth. This is called dietary restriction — what you eat.
Alternate between glucose and fats as food source
Similarly, let’s look at why alternation between glucose and fats promotes longevity. The major biological pathway to look into here is insulin signaling.
Insulin signaling
- Definition: insulin signaling is a pathway that regulates how our tissues maintain fuel homeostasis, i.e., the balance of energy expenditure and energy intake. It does so by controlling insulin levels and adjusting insulin sensitivity, i.e., our cells’ sensitivity in response to insulin. In simple words, if you have a high energy intake (overeating) and a low energy expenditure (lack of physical activity), your pancreas will pump a lot of insulin into the blood but your cells will not have the need for fuel and, thus, become insulin resistant and you will gain weight. The next step is diabetes type 2.
- Relationship to longevity: In general, you want to have better insulin signaling and higher insulin sensitivity to live longer. This translates into lower glucose and insulin levels. Studies on mice, rats, and humans have shown that glucose/carbohydrate restricted diets (including ketogenic) lead to lower insulin and increase lifespan. For example, one study has demonstrated that low carbohydrate diets administered to mice starting at middle age leads to 10%+ increase in lifespan compared to the control group (65% kcal from carbohydrates). That’s 8 more years of lifespan for an average human who switches to a ketogenic-like diet at 40 years old assuming we can directly translate results from mice to humans. As an added benefit, glucose restricted diets decrease cancer risks. Conversely, elevated blood glucose levels lead to formation of advanced glycation end products (aka AGEs) — one of the major biomarkers of aging and late onset diseases.
- Best way to improve/upregulate: using mostly ketones for fuel instead of glucose which means eating less, eating less frequently, and eating less/no simple sugars (sweetened drinks, cakes, salad dressings, ripe bananas, grapes, etc.) and simple starches (white bread, refined flour products, cereal, etc.).
So it sounds like — to live longer we have to fast more and follow a low carbohydrate/ketogenic diet and not use glucose as fuel? Not so fast. Being in constant ketosis has been linked to insulin resistance in ratsand mice but this might translate to humans too. It seems that our bodies need to sense glucose periodically to secrete insulin and stay sensitive to it. This is called metabolic flexibility.
Important caveat — I’m not suggesting that you should eat simple sugars and simple starches. But don’t limit yourself in healthy vegetables (mostly cruciferous and leafy greens) and make sure to regularly consume resistant starch which may drop you out of ketosis temporarily.
The “Why”: what’s often wrong and why
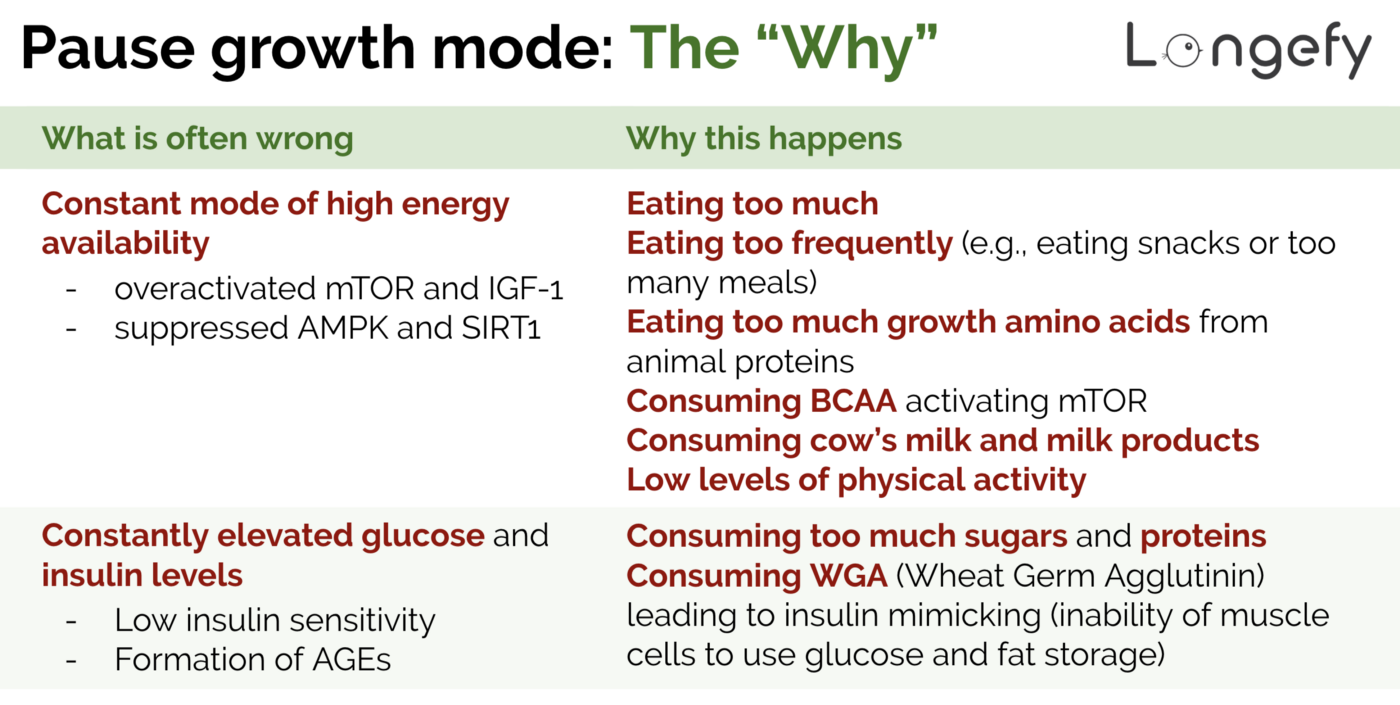
Cycle growth-regression and glucose-fats — sounds easy enough. Unfortunately, that’s not how most of us live. Not because we eat too much or too often (though that’s also one of the reasons) but because we, often unknowingly, eat certain life-shortening products.
Here are the key reasons why we are in the constant growth mode and never in the regression mode:
- We eat too much and have low levels of physical activity. While consuming more energy than spending leads to inhibition of growth hormone and results in obesity, it leads to IGF-1 activation and decrease in insulin sensitivity as this study demonstrates.
- We consume too much animal proteins. Animal proteins contain a lot of growth amino acids — methionine, leucine, and isoleucine — which activate the mTOR pathway.
- We consume BCAA. If you’re into fitness, you probably want to grow muscles and might be supplementing with BCAA. While they help grow muscles, they will definitely age you quicker by activating the mTOR pathway and promoting oxidative stress and inflammation. If you’re optimizing for overall health and lifespan, it’s best to avoid BCAA.
- We consume cow’s milk and milk products. Cow’s milk and milk products contain IGF-1, especially from cows treated by recombinant bovine growth hormone (rBGH), which is present in the milk even after pasteurization. While there is contradicting evidence in studies, if cow’s milk and milk products lead to cancer, they do lead to IGF-1 activation. As discussed, you want to inhibit exogenous IGF-1 to age less.
In addition, we often have elevated glucose levels and low insulin sensitivity because:
- We consume too much sugars and proteins. It has been shown that consuming high amounts of sugars (fructose, galactose, sucrose, sugar starch, etc.) decreases insulin sensitivity. It’s very easy to consume high amounts of sugar — a 20oz (500ml) bottle of Coke has 65 grams of sugar which is ~2 times higher than the recommended maximum daily intake of sugar by the American Heart Association. Even this recommendation is likely too high — Dale Bredesen, a leading researcher on Alzheimer’s disease, in his book “The End of Alzheimer’s”mentions that our bodies can safely process at most 15 grams of sugar per day. High protein consumption (>0.83g of protein per kg of body weigh per day) has also been linked to insulin resistance. If your weight is 80kg (176 lbs), then anything over 66 grams of protein a day (0.83 grams by 80 kg) can be considered a high protein diet leading to insulin resistance. A single burger a day will give you 58 grams of protein and I bet that’s not the only meal most people have a day. So the majority of us consume way too much protein.
- We consume WGA (aka Wheat Germ Agglutinin). WGA is a lectin that protects wheat from insects, yiest, and bacteria. As the name suggests, WGA can be found in wheat products (grain products, cereal, etc.). In short, WGA binds to muscle insulin receptors (mimicking insulin action) and blocks glucose from accessing muscle cells. WGA can also bind indefinitely to fat cells and instruct them to store glucose as fat. Overall, WGA decreases insulin sensitivity due to its insulin mimicking action and leads to elevated blood glucose levels. If you’re interested to learn more, here is an interesting deep-dive into dangers of lectins and WGA.
Clearly, this is a not a great recipe for a long and disease free life.
The “How”: what you can practically do and how it minimizes aging
The recipe to fix these issues is quite simple. Let’s zoom into nutrition, supplements, and physical activity recommendations.

Nutrition
- Practice caloric restriction — eating 20–40% fewer calories than you normally need — as often as you can.Caloric restriction (CR) is universally accepted as one of the best ways to extend lifespan. In this 20-year study on monkeys, CR improved life-expectancy and reduced the incidence or delayed the onset of age-associated pathologies. While it’s not feasible to run studies on CR impact on human lifespan, at least a single study on CR in humansdemonstrated decreased markers of aging such as inflammation. Judging by the animal studies, if a 48-year old man starts on a 30% calorie restriction, his normal life expectancy of 78 would be increased by ~3 years.
- Practice short-term (24/48/72 hour fast) and/or intermittent fasting/time-restricted eating.
Being on a constant 30%+ caloric restriction is tough for practical reasons — partly because food is one of the greatest pleasures. That’s why the best bet is to incorporate short-term fasts and time-restricted eating (aka intermittent fasting) into your schedule. This inhibits mTOR and activates autophagy. If you do a longer fast (24+ hours), it might be a good idea to work out on those fasting days to prevent muscle loss as Peter Attia does. - Practice a modified keto diet to maintain a state of mild ketosis by: 1) eating healthy fats (avocado, nuts, olive oil, MCT oil, etc.) and vegetables (cruciferous, leafy greens, resistant starch, etc.); 2) limiting unhealthy carbs (simple sugars and simple starches); 3) eating within a 12-hour window or less (time-restricted eating).As discussed above, being in ketosis has many benefits to insulin sensitivity and metabolic health but being in constant ketosis may decrease insulin sensitivity. In addition, consuming healthy vegetables is important for gut microbiota while a ketogenic diet may be harmful for composition of gut microbiota.
- Limit consumption of red meat and poultry to minimize consumption of growth amino acids: 1) Red meat: once a week or less; stick to grass-fed and -finished beef and heritage pork; 2) Poultry (chicken, etc.): a little bit every day or every other day; stick to pasture raised.Dr. Steven Gundry, a former cardiac surgeon who now specializes on nutritional medicine, in his book “The Longevity Paradox” recommends consuming a maximum 4 ounces of red meat once a week and 4 ounces of poultry once a day.
- Avoid consumption of cow’s milk and milk products — too much IGF in addition to having casein A1 which is poorly digested by humans. Switch to goat or sheep’s milk.
We already discussed the importance of avoiding exogenous IGF-1 for anti-aging purposes. Another point to keep in mind is that most milk on the market comes from A1 casein cows. When digested, casein A1 leads to production of a protein called beta-casomorphin-7 (BCM-7). It has been linked to autism, diabetes, and other diseases. If you like milk, use goat or sheep’s milk instead of cow’s milk because it has casein A2 and up to 5 times less IGF-1.
Supplements
- Moderate cow whey protein consumption to one serving a day or less— and only grass-fed and A2 casein.
Too much cow’s whey protein is bad because it upregulates the mTOR pathway. Consume only moderate amounts of whey proteins to not exceed the maximum recommended daily intake of proteins of 0.83 grams per kg of body weight per day. I consume one serving a day (21 g) and get the rest of my daily protein needs (45 grams) from collagen protein, nuts, eggs, and fish.
Importantly, you want whey protein from grass-fed cows and breeds producing A2 casein (because small amount of casein still pass into the whey powder and this can be harmful). Here is my favorite brand from grass-fed Jersey breed. The majority of whey proteins are artificially flavored with synthetic sweeteners such as aspartame and sucralose. They kill the good microbes in your gut. Look for brands with no flavors or sweeteners.
You can also switch to goat/sheep whey protein which has been shown to protect from mTOR overactivation.
Lastly, can you consume plant based proteins to get the benefits of whey protein? Unfortunately, not. Amino acids (specifically, cysteine) in whey protein boost production of the master antioxidant called glutathione. It improves liver detox, fights off free radicals, boosts your energy, and much more. Cysteine in good amounts is only found in whey protein; plant-based proteins have almost zero cysteine so consuming them wouldn’t help with glutathione synthesis. - Avoid BCAA. You can get enough amino acids from healthy foods (wild fish, nuts, eggs, etc.) and whey, collagen, and plant-based proteins.
- Supplement with exogenous ketones (MCT oil). MCT oil can bring you back into the state of mild ketosis in case you ate high carb meals such as resistant starch. Consuming MCT oil has been documented to have many benefits including improved insulin sensitivity and lower cholesterol levels.
- Consider supplementing with SIRT1 activators (Resveratrol, Fisetin, Quercetin, Butein, etc.), AMPK activators (Metformin, Berberine, Oxaloacetate, etc.), and NAD+ boosters (NMN, NR, etc.). Each compound is worth a separate deep-dive which I’ll do in later articles.
Physical activity
- Do moderate amounts of physical activity every day — walking, moving, running, swimming, strength training, etc.
All types of physical activity can improve health and lifespan via different mechanisms. Endurance exercise (swimming) has been linked with enhanced SIRT1 activity and compensation of age-associated decline in IGF-1 levels. Exercise also activates AMPK and improves NAD+ availability. While resistance exercise activates mTOR, it only happens in the skeletal and hearth muscles and the brain while being inhibited mTOR in liver and fat which may underlie some of the health benefits in these tissues.
In practice
I regularly practice extreme caloric restriction for overall health benefits. This includes eating only one 650–700 kcal meal a day.
Extreme caloric restriction has been proven to have extreme benefits. A study published in 2011 showed that extreme caloric restriction (600 kcal per day) for eight weeks reversed diabetes type 2 in 100% of patients. Another study published in 2019 with the same protocol (600 kcal per day for 8 weeks) reversed diabetes in ~80% of patients. Even 12 months after returning to a regular diet 30% of patients remained diabetes free.
While I don’t do extreme caloric restriction for 3 months in a row, I do it at least 1–2 times a week eating 650–700 kcal per day. Here’s what my meal looks like on those days:

I build this healthy meal myself from an otherwise not very healthy salad bar in the office. You just have to know what to pick. This meal consists of:
- One avocado (~250 calories)
- Soaked walnuts, 10 halves (~150 calories)
- Steamed broccoli, 150gr (~50 calories)
- Steamed brussels sprouts, 150gr (~50 calories)
- Olive oil, 15ml (~130 calories)
- Blackberries, 75gr (~30 calories)
- Collard greens, 100gr (~30 calories)
